Membrane Trafficking, Ubiquitin and Signalling
Sébastien LEON
 Our team seeks to determine the adaptation strategies of cells to nutritional stresses, such as nutrient deficiencies or inhibition of metabolism. Our theme is at the intersection of cell biology, cell signaling and metabolism, using the yeast Saccharomyces cerevisiae as a model.
Our team seeks to determine the adaptation strategies of cells to nutritional stresses, such as nutrient deficiencies or inhibition of metabolism. Our theme is at the intersection of cell biology, cell signaling and metabolism, using the yeast Saccharomyces cerevisiae as a model.
Keywords: Endocytosis, Membranes, Trafficking, Nutrient transporters, Glucose metabolism and signaling, AMPK / Snf1, Ubiquitin, Nedd4 / Rsp5, Arrestin-related proteins (ARTs, ARRDCs), Yeast, Cellular imaging, Proteomics, Cancer metabolism, 2-deoxyglucose
+33 (0)1 57 27 80 57 Contact
Our team studies the adaptation strategies of cells to nutritional stresses, such as nutrient deficiencies or inhibition of metabolism. Our work has contributed to the understanding of the mechanisms governing nutrient transporter endocytosis in the face of glucose fluctuations. This involves proteins of the arrestin family, which are regulated by signaling pathways in response to nutritional changes. More recently, we have initiated a new theme by studying the cellular consequences of a metabolic inhibitor, 2-deoxyglucose, on trafficking and we seek to understand the mechanisms of resistance to this drug.
1/ Regulation of endocytosis by the nutritional status of the environment
Endocytosis is a fundamental cellular mechanism that regulates the presence and abundance of proteins (receptors, transporters) at the plasma membrane.
Endocytosis of receptors is usually induced by the presence of the corresponding ligand (hormone, growth factor) in the extracellular medium and contributes to limit the exposure of cells to the signal. Defects in endocytosis can lead to aberrant signaling and be at the origin of pathologies including certain cancers.
Endocytosis also regulates the presence of nutrient transporters at the plasma membrane according to the nutritional composition of the environment and its possible changes. It is therefore an important component of the adaptation of cells to their environment.
Endocytosis of transporters is usually triggered by their ubiquitylation, which acts as a molecular signal. In yeast, transporter ubiquitylation is performed by Rsp5, a ubiquitin ligase of the Nedd4 family, which has 9 members in humans. This step also involves adaptor proteins called ARTs (Arrestin-Related Trafficking adaptors). In humans, ARTs-like proteins (called ARRDCs) appear to function in a similar way, and act as tumor suppressors.
Our goal is to understand how extracellular signals control arrestin function, and therefore transporter ubiquitylation and endocytosis during cell adaptation to a changing environment.
Our current model organism is the yeast Saccharomyces cerevisiae, which has contributed greatly to the understanding of conserved cellular mechanisms, including endocytosis
This project is situated in the context of the adaptation of cells to the presence or absence of glucose in the surrounding environment. Indeed, changes in glucose availability lead to profound changes in the landscape of transporters present at the plasma membrane. For example, glucose deficiency triggers endocytosis of most glucose transporters (e.g. Hxt3, Figure 1). Conversely, the addition of glucose to cells grown in the presence of alternative carbon sources leads to endocytosis of the corresponding transporters (e.g. Jen1, Figure 1 and Movie 1), to ensure that glucose is the preferred carbon source for use.
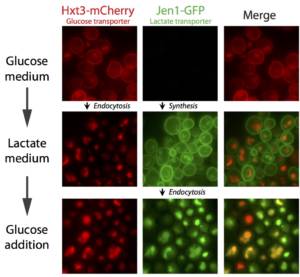
Figure 1. Glucose removal or addition trigger the endocytosis of selected transporters. © Sébastien LEON, CNRS/IJM
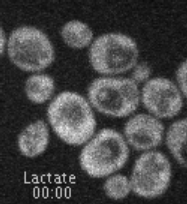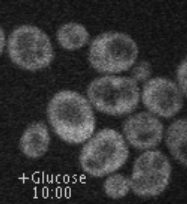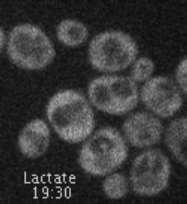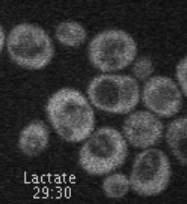
Movie 1. Glucose-induced endocytosis of the lactate transporter, Jen1. Yeast cells expressing Jen1-GFP were perfused with medium containing lactate (10’) and then glucose (45’) using a microfluidics device, and are imaged sequentially. Jen1-GFP departs from the plasma membrane within minutes after glucose treatment, to reach endosomal compartments and later, the large lytic compartment called vacuole, the yeast equivalent of lysosomes.
(ImagoSeine imaging facility, IJM). © Sébastien LEON, CNRS/IJM ]
In our lab, we delineated some of the molecular mechanisms at stake in these regulations. In particular, we found that nutrient signaling pathways modulate the post-translational modifications of ART proteins and their activity. The regulation of ART activity by nutrient availability explains why transporter ubiquitylation and endocytosis is triggered only in a specific physiological condition.
Our work has recently extended to the effects of a metabolic inhibitor, 2-deoxyglucose, on endocytosis.
2/ Toxicity of a metabolic inhibitor, 2-deoxyglucose, and associated resistance mechanisms
2-Deoxyglucose (2DG) is a glucose analog that inhibits cell growth by interfering with glucose utilization in many cellular processes, including glycolysis and protein N-glycosylation. 2DG has been considered for the treatment of tumors, following observations that the majority of cancer cells have increased glucose consumption. A glucose analogue, 2-fluoro-2-deoxy-glucose, is used to detect certain tumors (PET-scans).
However, some cells are able to bypass the toxicity of 2DG. We are investigating the mechanisms of resistance to this inhibitor through genetic screens in yeast, based on the characterization of mutants that have spontaneously become resistant to 2DG and the sequencing of their genome. Our work showed that 2DG triggers the activation of several signaling pathways and the induction of enzymes detoxifying a 2DG-derived metabolite. A similar enzyme has been identified in humans.
Other resistance strategies are currently under investigation.
Members
 Véronique ALBANESE, Chercheur, LEON LAB+33 (0)1 57 27 80 50, room 355B
Véronique ALBANESE, Chercheur, LEON LAB+33 (0)1 57 27 80 50, room 355B Alberto BALLIN, Doctorant, LEON LAB+33 (0)1 57 27 80 50, room 355B
Alberto BALLIN, Doctorant, LEON LAB+33 (0)1 57 27 80 50, room 355B Alexandre CORNILLE, Doctorant, LEON LAB+33 (0)1 57 27 80 50, room 355B
Alexandre CORNILLE, Doctorant, LEON LAB+33 (0)1 57 27 80 50, room 355B Samia MILED, ATER, LEON LAB+33 (0)1 57 27 80 57, room 355B
Samia MILED, ATER, LEON LAB+33 (0)1 57 27 80 57, room 355B Margot MOMEY, Stagiaire, LEON LAB+33 (0)1 57 27 80 50, room 355B
Margot MOMEY, Stagiaire, LEON LAB+33 (0)1 57 27 80 50, room 355B Matthieu SANIAL, Assistant ingénieur en biologie, LEON LAB+33 (0)1 57 27 80 50, room 355B
Matthieu SANIAL, Assistant ingénieur en biologie, LEON LAB+33 (0)1 57 27 80 50, room 355B Valerie SERRE, Enseignant-chercheur, LEON LAB+33 (0)1 57 27 80 50, room 355B
Valerie SERRE, Enseignant-chercheur, LEON LAB+33 (0)1 57 27 80 50, room 355B
To contact a member of the team by e-mail: name.surname@ijm.fr
Kahlhofer J, Leon S, Teis D, Schmidt O. The alpha-arrestin family of ubiquitin ligase adaptors links metabolism with selective endocytosis. Biology of the cell. 2021;113(4):183-219. Epub 2020/12/15. doi: 10.1111/boc.202000137. PubMed PMID: 33314196.
Le Boulch M, Brossard A, Le Dez G, Leon S, Rabut G. Sensitive detection of protein ubiquitylation using a protein fragment complementation assay. J Cell Sci. 2020;133(12):jcs.240093. doi: 10.1242/jcs.240093. PubMed PMID: 32409563.
Laussel C, Leon S. Cellular toxicity of the metabolic inhibitor 2-deoxyglucose and associated resistance mechanisms. Biochem Pharmacol. 2020;182:114213. doi: 10.1016/j.bcp.2020.114213. PubMed PMID: 32890467.
Ivashov V, Zimmer J, Schwabl S, Kahlhofer J, Weys S, Gstir R, Jakschitz T, Kremser L, Bonn GK, Lindner H, Huber LA, Leon S, Schmidt O, Teis D. Complementary alpha-arrestin-ubiquitin ligase complexes control nutrient transporter endocytosis in response to amino acids. eLife. 2020;9. doi: 10.7554/eLife.58246. PubMed PMID: 32744498.
Defenouillėre Q, Verraes A, Laussel C, Friedrich A, Schacherer J, Leon S. The induction of HAD-like phosphatases by multiple signaling pathways confers resistance to the metabolic inhibitor 2-deoxyglucose. Sci Signal. 2019;12(597):aaw8000. doi: 10.1126/scisignal.aaw8000. PubMed PMID: 31481524.
Hovsepian J, Albanese V, Becuwe M, Ivashov V, Teis D, Leon S. The yeast arrestin-related protein Bul1 is a novel actor of glucose-induced endocytosis. Mol Biol Cell. 2018;29(9):1012-20. doi: 10.1091/mbc.E17-07-0466. PubMed PMID: 29514933; PMCID: PMC5921569.
Hovsepian J, Defenouillere Q, Albanese V, Vachova L, Garcia C, Palkova Z, Leon S. Multilevel regulation of an alpha-arrestin by glucose depletion controls hexose transporter endocytosis. J Cell Biol. 2017;216(6):1811-31. Epub 2017/05/05. doi: 10.1083/jcb.201610094. PubMed PMID: 28468835; PMCID: PMC5461024.
Becuwe M, Leon S. Integrated control of transporter endocytosis and recycling by the arrestin-related protein Rod1 and the ubiquitin ligase Rsp5. eLife. 2014;3:03307. doi: 10.7554/eLife.03307. PubMed PMID: 25380227; PMCID: 4244573.
Becuwe M, Vieira N, Lara D, Gomes-Rezende J, Soares-Cunha C, Casal M, Haguenauer-Tsapis R, Vincent O, Paiva S, Léon S. A molecular switch on an arrestin-like protein relays glucose signaling to transporter endocytosis. J Cell Biol. 2012;196(2):247-59. Epub 2012/01/18. doi: 10.1083/jcb.201109113. PubMed PMID: 22249293.
Leon S, Erpapazoglou Z, Haguenauer-Tsapis R. Ear1p and Ssh4p are new adaptors of the ubiquitin ligase Rsp5p for cargo ubiquitylation and sorting at multivesicular bodies. Mol Biol Cell. 2008;19(6):2379-88. Epub 2008/03/28. doi: 10.1091/mbc.E08-01-0068. PubMed PMID: 18367543.
Publications
Revues
- Alberto BALLIN (en cours, 2021-XXXX)
- Clotilde LAUSSEL (2021) “Mécanismes de résistance et de toxicité à l’inhibiteur métabolique, le 2-deoxyglucose”
- Junie HOVSEPIAN (2017) “L’arrestine Csr2/Art8 régule l’endocytose des transporteurs d’hexoses lors d’une carence en glucose chez Saccharomyces cerevisiae”
- Michel BECUWE (2014) “Rôle et régulation de l’Arrestine Rod1/Art4 dans le trafic des transporteurs chez la levure Saccharomyces cerevisiae”
- Alexandre SOULARD, Université Lyon I
- France Gwenaël RABUT, IGDR Rennes
- France Florian FROEHLICH, Univ. Osnabrück, Allemagne
- David TEIS, BioZentrum Innsbruck, Autriche
- Helle ULRICH, IMB Mainz, Allemagne
Our work has been funded by the Agence Nationale pour la Recherche (ANR), the Fondation ARC pour la Recherche sur le Cancer, the Ligue contre le Cancer (Comité de Paris), the Fondation pour la Recherche Médicale (FRM), the Université de Paris (IDEX), and the CNRS.


